Proteins, Explained
You’ve heard the word protein before. On a food label, in a biology class, from someone at the gym who will not stop talking about their macros. But strip away all of that and here’s what protein actually is: the molecule your body uses to do almost everything.
Every reaction happening in your cells right now, every signal your body is sending, every structure holding your organs in place; there’s a protein behind it. You are, in a very literal sense, a machine held together and operated by tiny folded chains called proteins. And understanding what those chains are, how they’re built, and why their shape matters so much is how you start to understand one of the most fundamental building blocks of biology, of life.
So. Let’s build a protein.
Start with Amino Acids
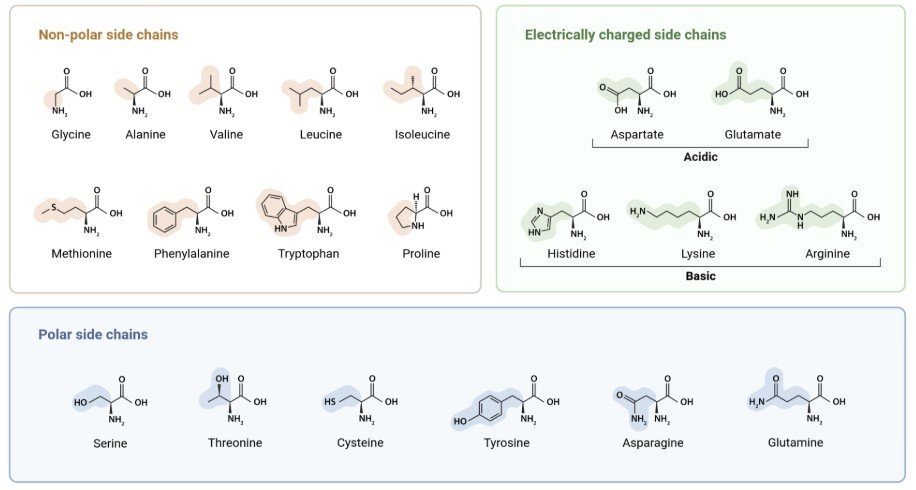
Proteins are built from amino acids. Think of amino acids as letters and proteins as words, the letters themselves are simple, but the order you put them in determines everything about the word you get.
There are 20 amino acids your body works with. Each one has the same basic backbone, but a different side chain: a little chemical tag hanging off it that gives it its personality. Some side chains are positively charged while some are negatively charged. Some are hydrophobic, meaning they actively avoid water. Some are hydrophilic, meaning they’re attracted to it. These properties matter a lot later.
Your DNA carries the instructions for which amino acids to string together and in what order. That instruction is then read by a ribosome, which links the amino acids one by one into a long chain called a polypeptide. However, at this point it’s not a protein yet. It’s just a chain.
How and why proteins fold
That polypeptide chain doesn’t stay as a string. The moment it starts coming off the ribosome, it begins to fold. And it doesn’t fold randomly; it folds into a specific, precise shape determined by the sequence of amino acids and the forces acting between them.
Those forces are:
- Hydrophobic interactions — remember those water-avoiding amino acids? Inside a cell, which is a watery environment, hydrophobic side chains want to get as far from water as possible. So they collapse inward, toward the center of the chain, away from the watery surroundings. This is actually one of the biggest drivers of folding.
- Hydrogen bonds — certain atoms in the chain carry small partial charges and are attracted to each other. These attractions form hydrogen bonds that pull parts of the chain toward each other and lock them into place.
- Electrostatic interactions — positively and negatively charged side chains attract each other. Like magnets, opposites pull together and lock regions of the chain into position.
- Van der Waals forces — tiny, weak attractions between atoms that are very close together. Individually almost nothing, but across a whole protein they add up.
All of these forces acting simultaneously push and pull the chain until it settles into its lowest energy state, the most stable shape it can take. That’s the fold.
The four levels of structure
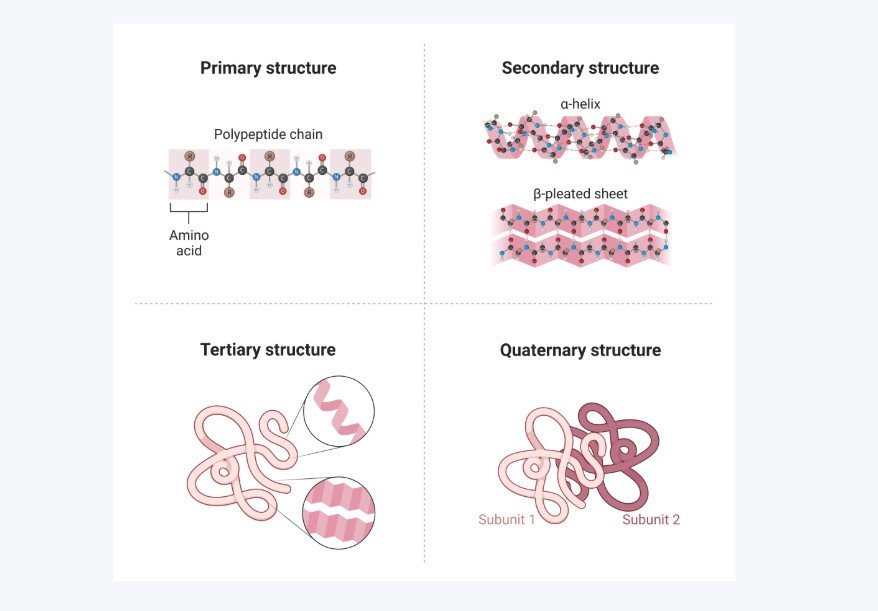
Biologists describe protein structure in four levels, each one building on the last.
- Primary structure — the sequence of amino acids itself. Just the order. This is determined entirely by your DNA.
- Secondary structure — as the chain folds, certain regions form repeating patterns. The two most common are the alpha helix, where the chain coils like a spiral staircase, and the beta sheet, where sections of the chain line up side by side and hydrogen bond together like a pleated ribbon. These form because of hydrogen bonding along the backbone of the chain.
- Tertiary structure — the full 3D shape of the whole polypeptide. This is where all those hydrophobic interactions, hydrogen bonds, and electrostatic forces come together to fold the chain into its final functional shape. This is what most people mean when they say “the protein.”
- Quaternary structure — some proteins are made of more than one polypeptide chain. When multiple chains come together to form one functional unit, that’s quaternary structure. Haemoglobin is a classic example, it’s made of four polypeptide chains working together.
What proteins actually do
Once folded, a protein can get to work. And the range of jobs they do is quite staggering.
Enzymes are probably the most important. An enzyme is a protein that speeds up a chemical reaction, sometimes by a factor of millions. Without enzymes, most of the chemistry keeping you alive would happen far too slowly to actually keep you alive. Your food wouldn’t get broken down fast enough. Your DNA couldn’t replicate. Your cells couldn’t produce energy. You’d essentially grind to a halt at the molecular level. Every single metabolic reaction in your body (and there are thousands happening simultaneously right now) depends on an enzyme.
Structural proteins hold things together physically. Collagen gives your skin, tendons, and bones their strength and flexibility. Keratin is what your hair and nails are made of. Actin and myosin are the proteins that make your muscles contract.
Transport proteins carry things around. Haemoglobin (a transport protein) carries oxygen from your lungs through your bloodstream to every cell in your body. Without it, your cells would suffocate.

Signalling proteins carry messages. Hormones like insulin are proteins; insulin signals to your cells that glucose is available and they should take it up. When that signal breaks down, you get diabetes.
Defense proteins fight infection. Antibodies are just proteins produced by your immune system that recognize and bind to specific foreign molecules, flagging them for destruction.
One molecule has five completely different jobs. And that’s not even all of them.
Why shape is everything, and what happens when it goes wrong
All of this, every single function listed above, depends entirely on the protein’s shape. An enzyme works because its shape creates a specific pocket called an active site that fits its target molecule like a lock and key. Change the shape, and the key no longer fits. The enzyme stops working.
So your cells take folding very seriously. There are dedicated proteins called chaperones (yes, actually called that) whose entire job is to help other proteins fold correctly and make sure they don’t clump together before they’re done. There are also quality control systems that identify misfolded proteins and tag them for destruction before they cause damage.
But sometimes things go wrong anyway. A random mutation in your DNA can change the amino acid sequence, thus changing the fold. Heat, chemical stress, or pH changes can disrupt the forces holding the shape together. Even worse, sometimes a protein just folds incorrectly for no clear reason.
And sometimes, rarely, but devastatingly, a misfolded protein doesn’t just stop working. It starts converting other proteins into its own misfolded shape. A chain reaction at the molecular level, spreading through tissue, impossible for your immune system to detect, and with no known cure.
That’s the next post.
For the visual learners:
>Amoeba Sisters – Protein Structure and Folding
>Professor Dave Explains – Protein Structure
>RCSBProteinDataBank – What is a Protein? Learn about the 3D shape and function of macromolecules
>Bozeman Science – Proteins